Draw The Lewis Structure For Chclo
Draw The Lewis Structure For Chclo - Determine the total number of valence electrons in the molecule or ion. Draw the lewis dot structure of chclo. Web draw the lewis structure for chclo. Web the lewis structure is a representation of the valence electrons in a molecule, which helps us understand the bonding and structure of the molecule.in this. Web lewis structure of hypochlorite (clo) a lewis diagram is a structural representation of valence electron distribution around an atom. Include all lone pairs of electrons. Find more chemistry widgets in wolfram|alpha. Web get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. The properly way to determine the lewis structure, based on this example, is: Draw the lewis structure for chclo. Experts have been vetted by. The properly way to determine the lewis structure, based on this example, is: A complex molecule's or ion's bond angle is the angle between the two. Web the ideal bonding angle(s) of the central atom of the compound of the chclo is 120°. Web lewis structure for hocl; Web draw the lewis structure for chclo. $7 + 6 + 1 = 14$ total. First, lets find the how many valence electrons chlorate has: Find more chemistry widgets in wolfram|alpha. How to draw a lewis structure of. Draw the lewis structure for chclo. A complex molecule's or ion's bond angle is the angle between the two. Find more chemistry widgets in wolfram|alpha. Web draw the lewis structure for chclo. Experts have been vetted by. Web draw out a correct lewis structure for the following compounds. There are 2 steps to solve this one. Draw the lewis dot structure of chclo. How to draw a lewis structure of. $7 + 6 + 1 = 14$ total. Draw the lewis structure for chclo. Include all lone pairs of electrons. Web draw the lewis structure for chclo. Draw the lewis dot structure of chclo. Experts have been vetted by. Include all lone pairs of electrons. First, lets find the how many valence electrons chlorate has: Experts have been vetted by. Draw the lewis structure for chclo. A complex molecule's or ion's bond angle is the angle between the two. Find more chemistry widgets in wolfram|alpha. The properly way to determine the lewis structure, based on this example, is: Experts have been vetted by. First, lets find the how many valence electrons chlorate has: $7 + 6 + 1 = 14$ total. There are 2 steps to solve this one. Draw the lewis dot structure of chclo. Web draw the lewis structure for chclo. Web draw out a correct lewis structure for the following compounds. Web lewis structure for hocl; How to draw a lewis structure of. Web it is possible to draw a structure with a double bond between a boron atom and a fluorine atom in bf 3, satisfying the octet rule, but experimental evidence indicates the bond. Web the lewis structure is a representation of the valence electrons in a molecule, which helps us understand the bonding. Determine the total number of valence electrons in the molecule or ion. Experts have been vetted by. Web draw the lewis structure for chclo. Web it is possible to draw a structure with a double bond between a boron atom and a fluorine atom in bf 3, satisfying the octet rule, but experimental evidence indicates the bond. A complex molecule's. Experts have been vetted by. Web get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Determine the total number of valence electrons in the molecule or ion. Draw the lewis dot structure of chclo. Draw the lewis structure for chclo. Web get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Draw the lewis structure for chclo. Determine the total number of valence electrons in the molecule or ion. How to draw a lewis structure of. Web it is possible to draw a structure with a double bond between a boron atom and a fluorine. Web get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Draw the molecule by placing the atoms on the grid and connecting them with bonds. First, lets find the how many valence electrons chlorate has: There are 2 steps to solve this one. How to draw a lewis structure of. Determine the total number of valence electrons in the molecule or ion. Draw the lewis structure for chclo. Web draw the lewis structure for chclo. Web draw out a correct lewis structure for the following compounds. Web the lewis structure is a representation of the valence electrons in a molecule, which helps us understand the bonding and structure of the molecule.in this. Include all lone pairs of electrons. $7 + 6 + 1 = 14$ total. Web lewis structure for hocl; Web the ideal bonding angle(s) of the central atom of the compound of the chclo is 120°. Web it is possible to draw a structure with a double bond between a boron atom and a fluorine atom in bf 3, satisfying the octet rule, but experimental evidence indicates the bond. Web steps for writing lewis structures.Draw The Lewis Structure For Chclo Drawing.rjuuc.edu.np
Draw The Lewis Structure For Chclo Drawing.rjuuc.edu.np
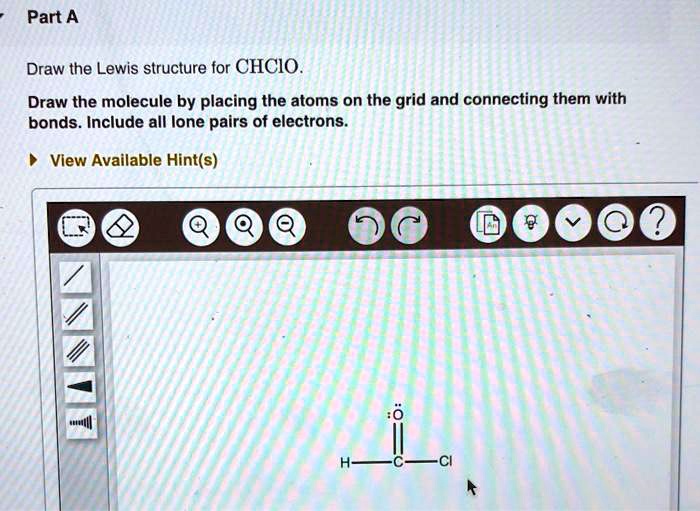
SOLVED Draw the Lewis structure for CHClO. Draw the molecule by
Chclo Lewis Structure What The Molecular Shape Of Chclo Clutch Prep
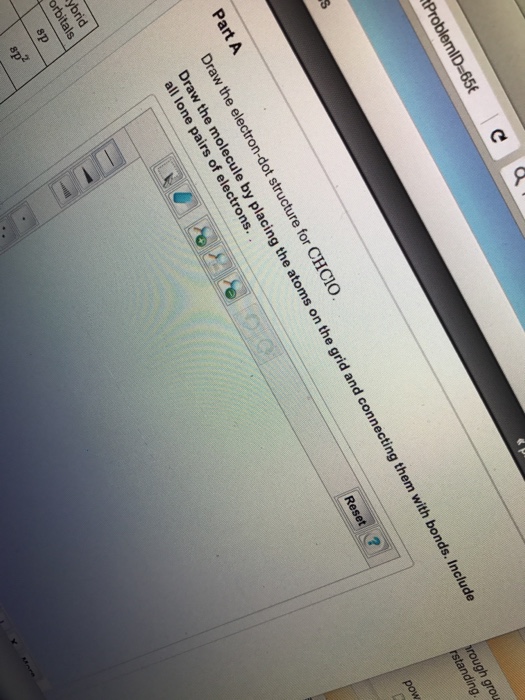
Solved Draw the electrondot structure for CHClO. Draw the
Chclo Lewis Structure What The Molecular Shape Of Chclo Clutch Prep
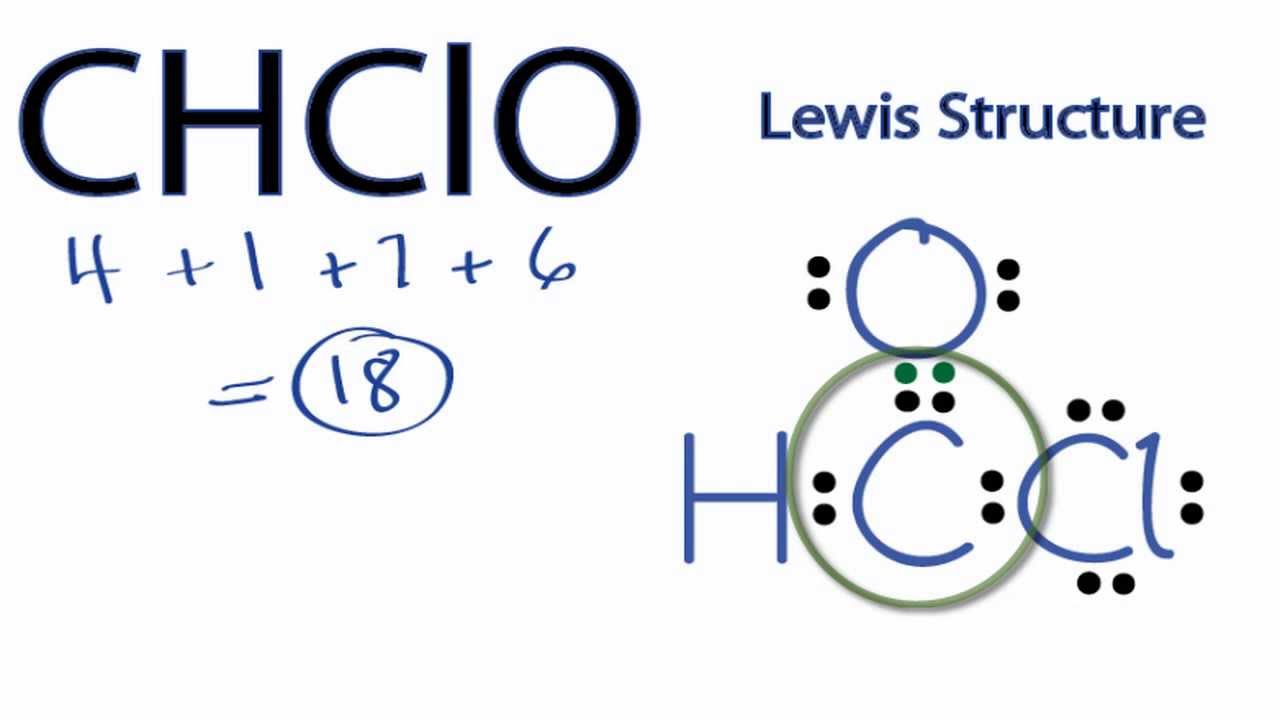
Lewis Structure of CHClO YouTube
CHClO Lewis Structure How to Draw the Lewis Structure for CHClO YouTube
Draw The Lewis Structure For Chclo Drawing.rjuuc.edu.np
Estructura De Puntos De Electrones Chclo
A Complex Molecule's Or Ion's Bond Angle Is The Angle Between The Two.
Draw The Lewis Dot Structure Of Chclo.
The Properly Way To Determine The Lewis Structure, Based On This Example, Is:
Find More Chemistry Widgets In Wolfram|Alpha.
Related Post: